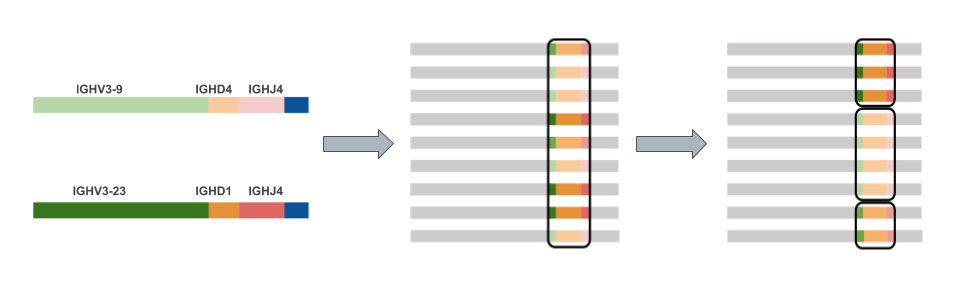 
Selected cells can revert back to the centroblast stage for more mutation and proliferation, or undergo a phase of proliferation, with daughter cells differentiating into either long-lived memory cells or antibody-secreting plasma cells. Centrocytes with high affinity for the antigen endocytose it, and present Ag peptides via MHC class II to follicular T H-cells, from which they receive further survival signals. Upon binding Ag complexes, the centrocytes may also receive further co-stimulatory signals from cell adhesion molecules on the FDCs, or secreted IL-6 and BAFF. Centrocytes, which may have Ab with altered Ag binding affinities, compete for a limited number of native antigen complexes retained on the surface of follicular dendritic cells (FDCs) via complement receptors 1 and 2, or FcRγII. By an unknown trigger, centroblasts differentiate into centrocytes, which downregulate Aicda expression, and upregulate membrane antibody (Ab) expression. The centroblasts, in addition to upregulating Aicda expression, also upregulate genes associated with a pro-apoptosis pathway ( 4). This allows precise lineage tracking among activated B-cells (centroblasts) that are clonally expanding, and acquiring Aicda-mediated gene mutations, within the follicle/germinal center ( 1– 3). VDJ recombination, which generates part of the unique antigen (Ag) binding site for the antibodies (Abs) produced by each B-cell, also provides a unique genetic ‘fingerprint’ for each B-cell. 4) Selected centrocytes receive survival signals from FDCs and follicular T H-cells that rescue them from a pro-apoptotic state. 3) post-mutation B-cells (now centrocytes) undergo a selection process that favors B-cells with improved antigen binding affinities. 2) During clonal expansion activation-induced cytidine deaminase (Aicda) generates somatic hypermutations in the immunoglobulin (Ig) VDJ exon of the B-cells. The germinal center (GC) process in mammals, which leads to antibody affinity maturation, has several hallmarks: 1) activated B-cells (plasmablasts) and T H-cells are recruited to the center by chemokines secreted by follicular dendritic cells (FDCs) and once there the B-cells (now centroblasts) clonally expand. These findings provide insights into the evolution of the affinity maturation process, the improvement of fish vaccines and possibly also the workings of atypical ectopic germinal centers generated in several human diseases. Melano-macrophages appear to trap the Ag used for post-mutation B-cell selection, performing a role analogous to the follicular dendritic cells of mammalian germinal centers. Finally, we show evidence for positive selection for replacement mutations in regions encoding the antigen contact loops, but not in the framework regions, consistent with functional antibody modification. Recruitment of B-cells to the clusters appears to be ongoing, as there are additional Ig clones having smaller lineages. Construction of Ig clonal lineage trees revealed that unlike surrounding lymphoid tissue, each cluster is dominated by a few B-cell VDJ clonotypes having hundreds of mutated variants. Sequenced IgH VDJ repertoire libraries from individual isolated clusters showed evidence of B-cell clonal expansion and VDJ somatic hypermutation. In fish there are clusters of Aicda + cells encircled by pigmented ‘melano-macrophages’ and we test the hypothesis that these clusters are functionally analogous to germinal centers. Cold blooded vertebrates lack germinal centers, yet have a functional Ig gene mutator enzyme, Aicda. Department of Biological Sciences, University of Alberta, BioSci Bldg, Edmonton, AB, CanadaĭNA mutagenesis during antibody affinity maturation has potentially oncogenic or autoimmune outcomes if not tightly controlled as it is in mammalian germinal centers.Doaa Waly *, Aradana Muthupandian, Chia-Wei Fan, Harrison Anzinger and Brad G.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
